Key Takeaways
- Shares of Definium Therapeutics climbed more than 13% during premarket hours following a presidential Executive Order supporting psychedelic medicine research
- The directive instructs the FDA to expedite research pathways and regulatory approval processes for psychedelic-based mental health therapies
- Company leadership responded enthusiastically, emphasizing their DT120 clinical program for GAD and MDD
- The stock has delivered returns exceeding 300% over the trailing twelve months, reaching a fresh high of $25.63
- Analyst consensus stands at Strong Buy with a collective price target of $38.75, suggesting approximately 70% upside potential
Shares of Definium Therapeutics experienced significant upward momentum Monday following President Donald Trump’s signing of an Executive Order designed to facilitate psychedelic drug research for mental health applications.
The presidential directive explicitly identified psychedelic compounds as promising therapeutic candidates for conditions such as depression, anxiety disorders, and suicide prevention. Additionally, the order mandated that the FDA eliminate regulatory obstacles currently impeding clinical investigation in this field.
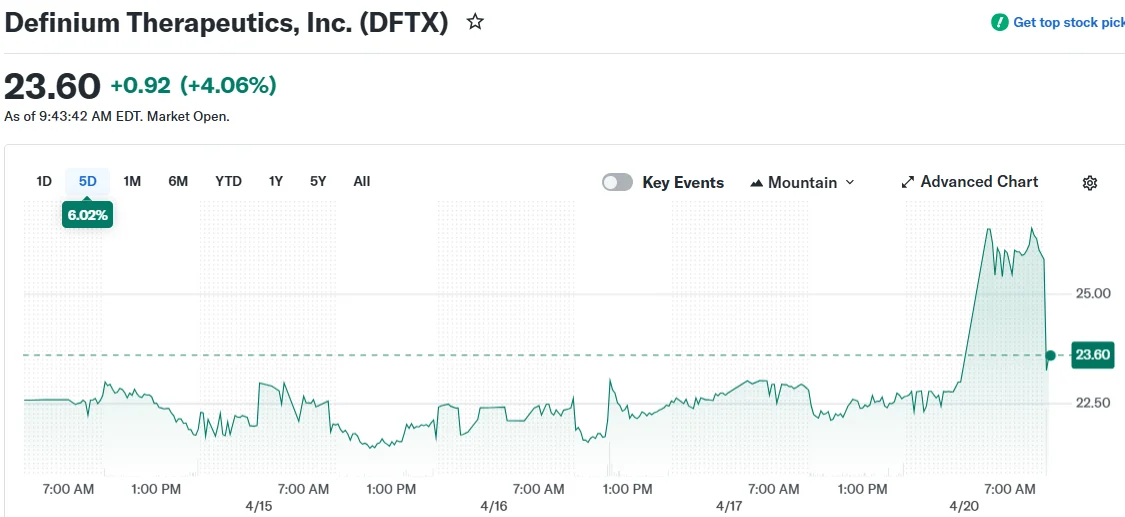
DFTX shares advanced 13.71% in premarket activity. The biotech stock has appreciated more than 300% across the past year, touching a new 52-week peak of $25.63 during the morning session.
Definium CEO Rob Barrow issued a strong statement welcoming the development. He expressed appreciation for “the Administration’s recognition that psychedelic medicines may represent meaningful new treatment options.” Barrow specifically highlighted DT120, the company’s lysergide tartrate compound currently in development for Generalized Anxiety Disorder and Major Depressive Disorder.
The White House order also addressed veteran suicide prevention, highlighting that approximately 6,000 veterans die by suicide annually across the United States. It noted elevated suicide rates during pandemic-related restrictions, which approached historical peaks. The directive positioned psychedelic therapeutics as a potentially effective intervention for these critical public health challenges.
Despite robust percentage gains in premarket activity, trading volume remained relatively subdued. Approximately 146,000 shares traded hands early in the session, well below the three-month average daily volume of roughly 1.68 million shares.
Wall Street Outlook and Price Projections
Analyst sentiment remains decidedly bullish on Definium. DFTX holds a Strong Buy consensus rating supported by 12 Buy recommendations issued within the last three months. The mean price objective stands at $38.75, implying approximately 70% appreciation from present trading levels.
Stifel launched coverage with a Buy recommendation, highlighting an elevated probability of clinical success for DT120 in treating GAD. Similarly, Piper Sandler initiated at Overweight, characterizing DT120 as a “best-in-class opportunity” within the emerging neuroplastogen therapeutic category.
Phase 3 clinical trial results for DT120 in Generalized Anxiety Disorder are anticipated during the first half of Q3 2026. The company’s next earnings announcement is scheduled for roughly 17 days from now.
Industry and Market Dynamics
Generalized Anxiety Disorder impacts more than 10% of American adults during any three-year timeframe, based on research published in the Journal of Mood and Anxiety Disorders. GAD prevalence across the United States reached 6.6% during 2023.
DFTX shares have gained 69.38% year-to-date, with the company maintaining a market capitalization near $2.26 billion.
According to InvestingPro analysis, the stock appears potentially undervalued relative to Fair Value calculations, though current RSI readings indicate overbought conditions. The stock’s beta coefficient of 2.56 signals elevated volatility relative to broader market benchmarks.
The 52-week low for DFTX was recorded at $5.45. Current trading activity places shares around $25.78.






